17. MCDC – AUSGEWÄHLTE BEITRÄGE
Poster: Evaluation of a novel air-liquid interface exposure system for the aerosolization of the chemical warfare agent sulfur mustard
Amelie Tsoutsoulopoulos*, Tanja Popp#, Markus Siegert*, Tabea Zubel+, Harald John*, Aswin Mangerich+, Annette Schmidt*,§, Harald Mückter‡, Thomas Gudermann‡, Horst Thiermann*, Dirk Steinritz*,‡
Introduction

As human exposure data are limited and translation of animal data to humans are difficult or even flawed due to critical differences (e.g. respiratory physiology and anatomy), recent research has focused on suitable in vitro exposure methods that represent a valuable alternative for explorative toxicological in vivo studies. Most exposure systems are thereby exposing cells under submerged conditions where the test substances are dissolved or suspended directly in the cell culture medium covering the cells. Such approaches may be flawed because they can affect and alter the physico-chemical properties of a compound and thus, the toxic properties of a test substance. Air-liquid interface (ALI) in vitro inhalation models, however, mimic the human exposure at a cellular level with higher biological and physiological similarity than submerged exposure experiments. So far, it is unclear, whether the enhanced experimental complexity of ALI exposure is superior to submerged exposures. Moreover, aerosolization of SM represents a -major challenge to scientists due to the extreme toxicity of SM, the safety precautions to be observed and the -homogenous distribution of the SM aerosol.
Aim
Aim of our study was the establishment, validation and application of an appropriate in vitro exposure system that meets all necessary safety requirements for the exposure of cultivated human lung cells to nebulized SM aerosols at the ALI. Furthermore, we analyzed whether exposing cells at the ALI is feasible and favorable compared to the classic submerse SM exposure.
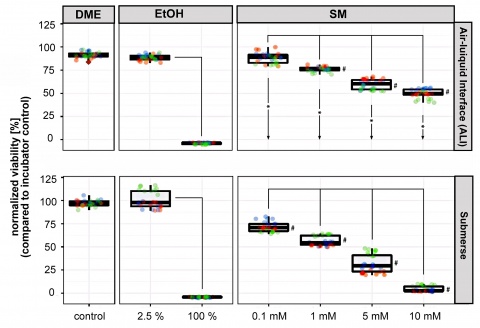 Figure 2: Viability of A549 cells after ALI and submerse SM exposure. Cell viability is expressed in % in relation to the incubator controls that were normalized
to 100 %. The upper boxplots represent the mean results of the cells exposed to SM under ALI conditions (4 exposure
experiments, each with 3 inserts per condition, resulting in n=12). The lower boxplots show the mean results of cells exposed under submerged conditions (3
exposure experiments, each with 3 inserts per condition, resulting in n=9). # indicates significant differences (p < 0.01) between SM concentrations and DMEM
control; lines indicate significant
differences (p < 0.01) between
concentration groups, and asterisks/
arrows between ALI and submerse groups.
Materials and Methods
Figure 2: Viability of A549 cells after ALI and submerse SM exposure. Cell viability is expressed in % in relation to the incubator controls that were normalized
to 100 %. The upper boxplots represent the mean results of the cells exposed to SM under ALI conditions (4 exposure
experiments, each with 3 inserts per condition, resulting in n=12). The lower boxplots show the mean results of cells exposed under submerged conditions (3
exposure experiments, each with 3 inserts per condition, resulting in n=9). # indicates significant differences (p < 0.01) between SM concentrations and DMEM
control; lines indicate significant
differences (p < 0.01) between
concentration groups, and asterisks/
arrows between ALI and submerse groups.
Materials and Methods

- Establishment of the exposure system (see Figure 1)
- Verification of the homogeneous distribution of the nebulized aerosol in our exposure system using Coomassie blue
- Exposure of human epithelial lung cells to SM aerosols (0.1-10 mM) under submerse or ALI conditions
- Determination and comparison (submerse / ALI) of cell viability after exposure using the WST-1 assay
- Detection of SM-specific DNA-adducts by antibody staining and mass spectrometry
Results
Viability of cells after ALI and submerse SM exposure
A549 cells were exposed to SM under submerged or ALI conditions. Exposure times and SM concentrations were kept identical between both exposure routes in order to compare the two experimental approaches. Exposure of cells to SM resulted in a dose-dependent decrease of cell viability. The cytotoxicity in ALI experiments was significantly lower compared to the submerged exposures at comparable SM concentrations (see Figure 2).
Qualitative detection of DNA-adducts after SM exposure by immunocytochemical staining using the 2F8 antibody
The fluorescence intensities of DNA-adducts were densitometrically evaluated by laser scanning microscopy. A concentration-dependent increase of the relative flu-orescence intensity after SM exposure was observed in both exposure groups, but was significantly higher in submerged exposed cells (see Figure 3B).
Horizontal lines in Figure 2B indicate background fluorescence levels, error bars represent standard deviations. The high standard deviation does not interfere with the homogeneity of the DNA-adducts. This is due to a distinct variation of fluorescence intensities because cell confluency was below 100 % and 2F8 staining was located in the nuclei.
Quantitative detection of DNA-adducts after SM exposure using μLC-ESI MS/HR mass spectrometry
SM-induced DNA alkylation was analyzed as a marker for cell damage by μLC-ESI MS/HR MS-based detection of HETE-Ade and HETE-Gua DNA-adducts. In line with the viability data and the immunocytochemical staining, increasing SM concentrations led to increasing amounts of DNA-adducts (HETE-Ade and HETE-Gua). Comparing the amount of DNA alkylation per 1x106 base pairs between ALI-exposed and submerse-exposed cells revealed a distinct higher DNA damage in submerged exposed cells (see Figure 4).
 Figure 4: Detection of DNA-adducts
after SM exposure using the μLC-ESI MS/HR MS. Amount of DNA damage per 1x106 bases increased with increasing
SM concentrations. Significant differences in DNA damage were measurable between cells exposed to SM under submerged conditions or at the ALI. Cells from three independent experiments (with three inserts per experiment) were pooled per SM concentration and exposure route (n=9). Error bars represent
standard deviations.
Conclusion
Figure 4: Detection of DNA-adducts
after SM exposure using the μLC-ESI MS/HR MS. Amount of DNA damage per 1x106 bases increased with increasing
SM concentrations. Significant differences in DNA damage were measurable between cells exposed to SM under submerged conditions or at the ALI. Cells from three independent experiments (with three inserts per experiment) were pooled per SM concentration and exposure route (n=9). Error bars represent
standard deviations.
Conclusion

Our results present the first in vitro exposure of cultivated human lung cells to nebulized SM aerosols under ALI conditions. We proved the safe applicability of our
ALI exposure system, and the reproducible and stable generation and homogenous distribution of the nebulized SM aerosol. Clear dose-response relationships can be derived. Lower doses of SM reached the cells during ALI exposure in our setup compared to the submerged -exposure but were sufficient to cause cytotoxicity and DNA-adduct formation. Our exposure system represents a scenario which is closer to the physiological conditions after an in vivo exposure to the agent compared to the in vitro submerged exposure.
References
- Tsoutsoulopoulos A, Siegert M, John H, Zubel T, Mangerich A, Schmidt A, Mückter H, Gudermann T, Thiermann H, Steinritz D, Popp T: A novel exposure system generating nebulized aerosol of sulfur mustard in comparison to the standard submerse exposure. Chem Biol Interact. 2019; 298: 121-128.
- Tsoutsoulopoulos A, Möhle N, Aufderheide M, Schmidt A, Thiermann H, Steinritz D: Optimization of the CULTEX® Radial Flow System for in vitro investigation of lung damaging agents. Toxicol Lett. 2016;244: 28-34.
- Steinritz D, Möhle N, Pohl C, Papritz M, Stenger B, Schmidt A, Kirkpatrick CJ, Thiermann H, Vogel R, Hoffmann S, Aufderheide M: Use of the Cultex® Radial Flow System as an in vitro exposure method to assess acute pulmonary toxicity of fine dusts and nanoparticles with special focus on the intra- and inter-laboratory reproducibility. Chem Biol Interact. 2013; 206: 479-490.
* Bundeswehr Institute of Pharmacology and Toxicology, Munich, Germany
# Bundeswehr Institute of Radiobiology, Munich, Germany
+ Molecular Toxicology Group, Department of Biology, University of Konstanz, Germany
§ Bundeswehr University Munich, Faculty of Human Sciences, Neubiberg, Germany
‡ Walther-Straub-Institute of Pharmacology and Toxicology, LMU, Munich, Germany
The poster was awarded the 1. price of the 17. MCDC poster competition.
For the authors
Captain (res.) Amelie Tsoutsoulopoulos, M. Sc.
E-Mail: [email protected]
Datum: 20.09.2019